Charging a Li–O2 Battery Using a Redox Mediator
The non-aqueous Li–air (O2) battery is receiving intense interest because its theoretical specific energy exceeds that of Li-ion batteries. Recharging the Li–O2 battery depends on oxidizing solid lithium peroxide (Li2O2), which is formed on discharge within the porous cathode. However, transporting charge between Li2O2 particles and the solid electrode surface is at best very difficult and leads to voltage polarization on charging, even at modest rates. This is a significant problem facing the non-aqueous Li–O2 battery.
Here in the article entitled “Charging a Li–O2 Battery Using a Redox Mediator” on Nature Chemistry (2013, 5, 489), the researchers found that incorporation of a redox mediator, tetrathiafulvalene (TTF), enables recharging at rates that are impossible for the cell in the absence of the mediator. Prof. Yongyao Xia was invited to write a review article entitled “An Agent for Change” for this finding by Nature Chemistry (2013, 5, 445).
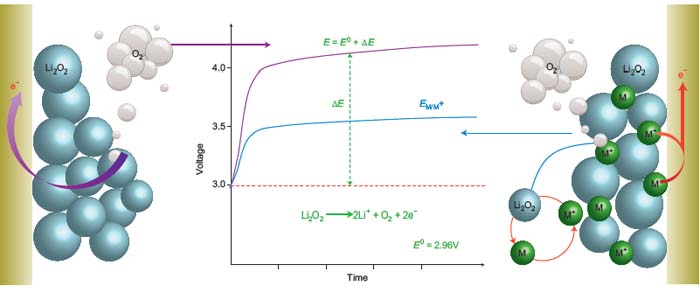
Figure: Schematic illustration of an electron–hole transfer agent that promotes the oxidation of solid Li2O2. Left: The electrochemical oxidation of Li2O2 (Li2O2→ 2Li+ + O2 + 2e–) is limited by the charge transport between the insulating Li2O2 particles and the electrode surface. Right: With a redox mediator (M) dissolved in the electrolyte to assist, this sluggish charge transfer is replaced by the free and fast diffusion of the mediator. M is directly oxidized at the electrode surface to M+ (M → M+ + e–), it then oxidizes the Li2O2 particles by a direct chemical reaction (2M+ + Li2O2 → 2Li+ + O2 + 2M), becoming itself reduced back to M. (E0, theoretical potential for Li2O2 oxidization; ΔE, polarization associated with charge transfer; E, potential at which Li2O2 is oxidized; EM/M+, electrochemical potential of oxdizing M).