Biomass derived Formic Acid as C1 source applied in N-methylation
Recently, iChEM researcher, Prof. Yao Fu from University of Science and Technology of China developed a non-noble metal catalysis system to catalyze the straightforward alkylation of amines with readily available carboxylic acids. The research result was published in Angew .Chem. Int. Ed. (Angew .Chem. Int. Ed., 2015, 54, 9042)
By using a boronic-acid catalyzed amide condensation, followed by B(C6F5)3-catalyzed reduction of amide and direct reductive N-methylation with formic acid, gave the drug molecule in 91 % yield (Figure 1)
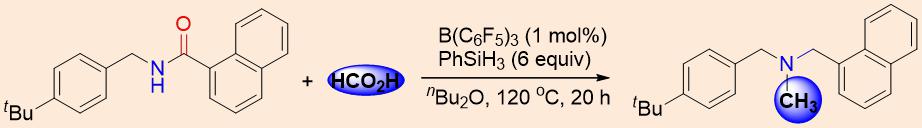
Figure 1. Synthesis of drug molecule Butenafine
Formic acid is an important platform chemical with wide application in medicine, spin, dyeing industry, and it is also an ideal C1 source derived from biomass. Fu group have developed an efficient method for biomass transformation to formic acid (ChemSusChem, 2012, 5, 1313), which provides a way for biomass utilization. Alkylated amine structures appear widely in pharmaceuticals, agrochemicals, and materials. Traditional methods of alkylating an amine include non-catalytic reactions using hazardous alkyl halides for substitutions, or using air-sensitive aldehydes for reductive aminations. Compared with traditional methods, Fu group developed a method for the alkylation of amines using carboxylic acid, which is more appealing because of the step economy and easy availability of the carboxylic acids.
Besides, they also studied the scope of carboxylic acids to explore the generality of this method. They also successfully applied biomass-derived levulinic acid to this transformation (Figure 2). It is noteworthy that this is the first report about Lewis acid [B(C6F5)3] that it can not only be applied in multi-bond reduction but also in C-N bond formation.
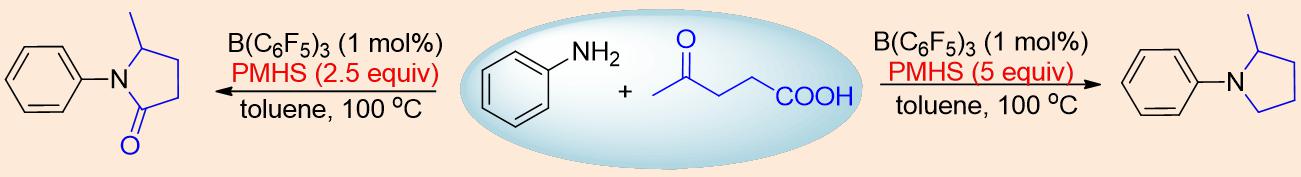
Figure 2. Levulinic acid applied in alkylation reaction
The first author of this paper is Mingchen Fu from University of Science and Technology (USTC). This work was supported by the 973 Program (2012CB215306), NSFC (21325208, 21172209, 21361140372) et al..
Link: http://onlinelibrary.wiley.com/doi/10.1002/anie.201503879/abstract