Organocatalytic, Asymmetric Total Synthesis of (-)-Haliclonin A
Recently, theiChEM Prof. Pei-Qiang Huang’s group first proposed a total synthesis of (-)-Haliclonin A and also confirmed the structure of natural (-)-haliclonin A. Their research result was published on the Angew. Chem. Int. Ed. (2016, 55, 10.1002/anie.201512005) as a title of “Organocatalytic, Asymmetric Total Synthesis of (-)-Haliclonin A”.
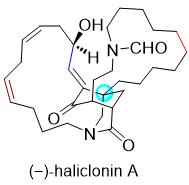
Figure 1. Structure of(-)-haliclonin A (1).
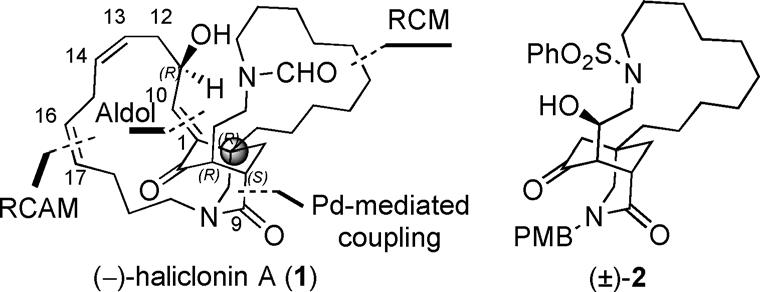
Figure 2. Key disconnections of (-)-haliclonin A (1) and a tricyclic core.
The macrocyclic alkaloid (-)-haliclonin A (1, Figure 1) was isolated in 2009 from a marine sponge Haliclona sp. collected from Korean waters by Shin and co-workers. The structure of 1 was determined using 2D NMR spectroscopy and mass spectrometry techniques. The absolute configuration of this compound remained confusing because it was partially assigned as 1E, 3S, 4R, 6S, 11S by Shin and co-workers on the basis of spectral and chemical analyses, but presented the enantiomer shown in Figure 1. (-)-Haliclonin A(1) contains two aza-macrocycles and is structurally related to sarains A–C, but possesses an unprecedented 3-azabicyclononane framework. Preliminary bioassays showed that this unique natural product exhibits moderate antibacterial activity against several microbial strains, and cytotoxicity against the K562 leukemia cell line. Despite its intriguing structure, no total synthesis of (-)-haliclonin A (1) has been reported to date. In connection with their interest in the total synthesis of alkaloids, they recently disclosed a racemic synthesis of a tricyclic core of (-)-haliclonin A (2, Figure 2). They have developed an asymmetric strategy for this skeleton and report the first total synthesis of (-)-haliclonin A (1). During the course of this work, new chemistry has been developed, which includes the thiourea 5-catalyzed asymmetric conjugate addition of nitromethane and 3-substituted cyclohex-2-enone, SmI2-mediated bimolecular reductive coupling of enone with aldehyde, and direct transformation of enol into enone.
This work was conducted under the guidance of Prof. Huang and accomplished by the Ph.D student Lian-Dong Guo and the graduate students Xiong-Zhi Huang and Wen-Sen Cao. The group members, Prof. Jian-Liang Ye and Prof. Yuan-Ping Ruan participated in the part of work for the project. This work was greatly supported by the NSFC, the 973 Program, the National Fundamental Scientific Talent Foundation, the MOE Cheung Kong Scholar Foundation and the State Key Laboratory of Applied Organic Chemistry in Lanzhou University.
Paper Link: http://onlinelibrary.wiley.com/doi/10.1002/anie.201512005/full