Environmentally-friendly aqueous Li (or Na)-ion battery with fast electrode kinetics and super-long life
Recently, the iChEM Prof. Yongyao Xia and Yonggang Wang’s group first proposed a new-type aqueous Li (or Na)-ion battery by using a polymer Li+ (or Na+) exchange membrane to separate a solid-state polymer anode and a liquid cathode containing water-soluble inorganic redox couples. Their research result was published on the Science Advances, a top international journal under Science, as a title of “Environment-friendly Aqueous Li (or Na)-ion Battery with Fast Electrodes Kinetics and Super-long Life”. 2014 Ph.D student Ms. Xiaoli Dong from Advanced Materials Laboratory of FDU is the first author. This article was overall evaluated as excellent and exciting and considered presenting an original battery system, and moreover, this battery displays inherent fast electrodes kinetics, long life and high safety which make this work quite important and innovative.
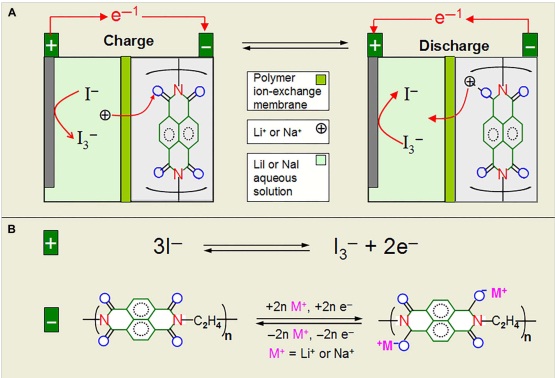
Current rechargeable batteries generally display limited cycle life and slow electrode kinetics and contain environmentally unfriendly components. Furthermore, their operation depends on the redox reactions of metal elements. Prof. Xia and Wang’s group present an original battery system that depends on the redox of I-/I3- couple in liquid cathode and the reversible enolization in polyimide anode, accompanied by Li+ (or Na+) diffusion between cathode and anode through a Li+/Na+ exchange polymer membrane. There are no metal element–based redox reactions in this battery, and Li+ (or Na+) isonly used for charge transfer. Moreover, the components (electrolyte/electrode) of this system are environment-friendly. Both electrodes are demonstrated to have very fast kinetics, which gives the battery a supercapacitor-like high power. It can even be cycled 50,000 times when operated within the electrochemical window of 0 to 1.6 V. Such a system might shed light on the design of high-safety and low-cost batteries for grid-scale energy storage. Such a system can exhibit a supercapacitor-like high-rate performance and a super-long cycle life because the kinetics of both electrode reactions are limited neither by the ion diffusion process nor by phase conversion. This finding not only provides a promising solution for grid-scale energy storage but also brings a new idea for battery design.
This work was greatly supported by the NSFC, the Science Committee of Shanghai and iChEM.
paper link:http://advances.sciencemag.org/content/2/1/e1501038