Ultrathin Co3O4 Layers Realizing Optimized CO2 Electroreduction to Formate
Most recently, iChEM researcher Prof. Yi Xie’s group made tremendous progress in “Ultrathin Co3O4 Layers Realizing Optimized CO2 Electroreduction to Formate”, which was published on Angew. Chem. Int. Ed., 2015, DOI: 10.1002/anie.201509800.
Global warming and the energy crisis are two important issues we are facing in the 21st Century. Both problems are rooted in the unsustainable utilization of fossil fuels accompanied with release of greenhouse gas CO2. In this regard, CO2 reduction into useful fuels, powered by a renewable electricity source, could not only help to reduce CO2 emission, but also decrease the energy shortage. Over the past three decades, researchers have evaluated lots of metals as electrodes for CO2 reduction in aqueous solutions. While these metal electrodes show competent CO2 electroreduction performances, they usually suffer from very high prices, limited availability, as well as rapid loss of CO2 reduction activity, which seriously hinder their large-scale practical applications. To address these issues, several naturally abundant and chemically stable transition-metal oxides have been recently explored as potential CO2 electroreduction catalysts to replace the expensive and easily deactivated metals. Among these materials, the spinel-type oxide of Co3O4 seems to be one of the most competitive candidates for electrocatalytic CO2 reduction. In addition to being environmental friendly, having low cost and an abundance of reserves, the Co3O4 adopts a normal spinel structure with Co2+ ions in tetrahedral interstices and Co3+ ions in octahedral interstices, in which the peculiar crystal structure enables it to be high environmental stability. In spite of these advantages, the catalytic CO2 reduction performance of Co3O4 is still far from satisfactory, which is primarily ascribed to the very low amount of active sites associated with poor electrical conductivity in previously fabricated Co3O4 catalysts. Henceforth, designing a suitable material with abundant active sites and high electrical conductivity holds the key to greatly promoting the electrocatalytic CO2 reduction performances.
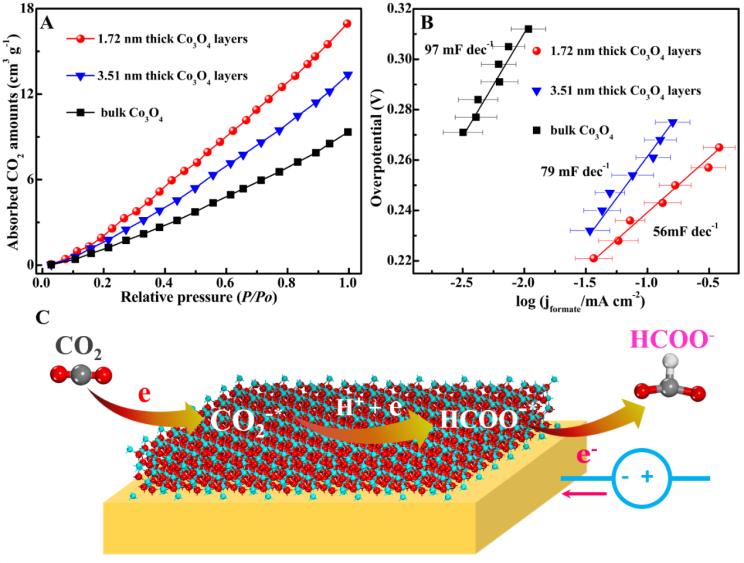
In their work, atomic layers for transition-metal oxides are proposed to address these problems through offering an ultralarge fraction of active sites, high electronic conductivity, and superior structural stability. As a prototype, 1.72 and 3.51 nm thick Co3O4 layers were synthesized through a fast-heating strategy. The atomic thickness endowed Co3O4 with abundant active sites, ensuring a large CO2 adsorption amount. The increased and more dispersed charge density near Fermi level allowed for enhanced electronic conductivity. The 1.72 nm thick Co3O4 layers showed over 1.5 and 20 times higher electrocatalytic activity than 3.51 nm thick Co3O4 layers and bulk counterpart, respectively. Also, 1.72 nm thick Co3O4 layers showed formate Faradaic efficiency of over 60 % in 20 h. This work was published on top international journal (Angew. Chem. Int. Ed) and selected as Hot Papers.
http://onlinelibrary.wiley.com/wol1/doi/10.1002/anie.201509800/full