To mitigate self-discharge of lithium–sulfur batteries by optimizing ionic liquid electrolytes
Recently, the iChEM researcher, Prof. Yongyao Xia’s group in FDU demonstrated pronounced suppression of polysulfide shuttle using an ionic liquid of electrolyte. The related work was published on Energy & Environmental Science as a title “Recent advances of pore system construction in zeolite-catalyzed chemical industry processes”.
Lithium–sulfur (Li–S) batteries are receiving tremendous attention due to their potential for high energy-density batteries in emerging electronics and vehicle applications. However, severe self-discharge associated with a rapid polysulfide redox shuttle process has remained a grand challenge, preventing the practical application of this attractive technology. Soluble polysulfide species (Li2Sx, 4 ≤ x ≤ 8) would continue to dissolve and migrate to the negative side because of concentration gradient, and then react with metallic Li, resulting in a decrease of open-circuit voltage, loss of upper discharge plateaus and discharge capacity. To address this issue, we take a different approach to the Li–S battery by developing a room temperature ionic liquid (RTIL) of N-methyl-N-propylpiperidinium bis(trifluoromethanesulfonyl)imide (PP13TFSI)-based electrolytes. When working together with LiNO3, zero self-discharge can be achieved to rest a full-charged cell for two days. From a systematic study of battery performance in terms of various kinds of electrolytes and comprehensive characterization on Li-electrodes, we conclude that both improvements on polysulfide diffusion control and stabilization of Li-metal should be fulfilled for a successful battery with low self-discharge. This approach shows the way forward to the practical use of Li–S batteries by selection and more rational design of electrolytes.
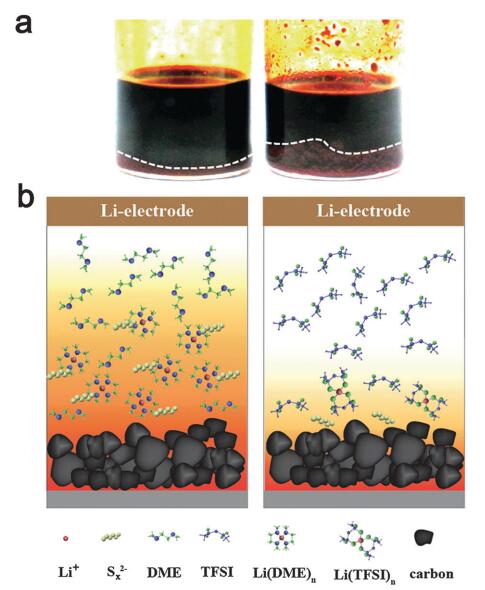
Li–S batteries generally suffer from severe self-discharge when resting due to an internal polysulfide shuttle effect. Soluble long-chain polysulfide species (Li2Sx, 4 ≤ x ≤ 8) would continue to dissolve and migrate to the negative side to react with metallic Li. They demonstrate pronounced suppression of polysulfide shuttle using an ionic liquid of the N-methyl-N-propylpiperidinium bis(trifluoromethanesulfonyl)imide (PP13TFSI)-based electrolyte. When working in combination with LiNO3, zero self-discharge can be achieved to rest a full-charged Li–S cell for two days. The fascinating study clearly demonstrates that a promising practical Li–S battery with low self-discharge depends on both improvements on polysulfide diffusion control and Li-metal stabilization. On the basis of the insight gained, they suggest the ultimate success of Li–S batteries depends on both improvements on polysulfide diffusion control and stabilization of Li-metal. Therefore, selection and design of electrolytes for Li–S batteries should fulfill both demands.
This work was financially supported by iChEM and NSFC.
http://pubs.rsc.org/en/content/articlepdf/2015/ee/c5ee02837j