Water-soluble molecules as co-catalysts to accelerate hole transfer
iChEM researchers, Prof. Yi Xie from School of Chemistry and Materials Science, USTC, collaborating with Prof. Qun Zhang, developed a novel method to make use of water-soluble molecules as co-catalysts to accelerate hole transfer for improved photocatalytic H2 evolution activity, which avoid the high cost of noble-metal co-catalysts. The result was published online in Nature Comm. ( Nature Comm. 2015, 6, 8647)
Photocatalytic conversion of solar energy to fuels is considered to be an ideal, renewable energy resource for the future, yet the relatively low-energy conversion efficiency remains the most critical factor that limits its practical applications. To achieve highly efficient photochemical conversion, the main challenge in photocatalysis is to maximize the extraction of charge carriers while suppressing their recombination. Noble-metal oxides are usually used as co-catalysts for hole transport because they can effectively lower the over potential for the oxidation reaction. Meanwhile, subjected to their high costs of upscaling, several cost-effective and earth-abundant alternatives have also been exploited and achieve relatively high efficiency.
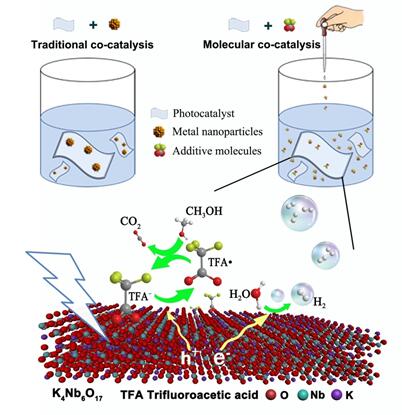
Fig. water-soluble molecules as co-catalysts to accelerate hole transfer for improved photocatalytic H2 evolution activity
Alternatively, homogeneous catalysis, free from limited contact areas, has gained considerable attention due to its high activity and selectivity. In this article, they propose a water-soluble molecular co-catalyst strategy to accelerate the hole transfer kinetics. The reversible redox couple TFA·/TFA–, making trifluoroacetic acid molecule (TFA) a robust molecular co-catalyst, provides further charge separation pathway across the catalysts interface, and consequently facilitates photocatalytic H2 generation. In situ electron spin resonance (ESR) together with energy-level alignment reveal that photo generated holes in K4Nb6O17 valence band readily react with the adsorbed TFA anions to yield TFA radicals and simultaneously the highly reactive radicals transfer holes to methanol, eliminating the main competitive pathway of electron–hole recombination. Moreover, ultrafast transient absorption spectroscopy combined with static as well as time-resolved photoluminescence spectroscopy confirms that effective charge separation is the primary factor behind the dramatically improved photocatalytic performance. Homogeneous co-catalyst, which is free from the limited contact areas between co-catalysts and reactants, provides sufficient active sites for catalysis, thus offering new opportunity to develop high-efficiency photosynthesis.
This work was financially supported by the National Basic Research Program of China, National Natural Science Foundation of China, Chinese Academy of Sciences, the Collaborative Innovation Center of Chemistry for Energy Materials and Collaborative Innovation Center of quantum information and quantum technology.
Link: http://www.nature.com/ncomms/2015/151021/ncomms9647/abs/ncomms9647.html