A new progress in photocatalytic water splitting via atomically controlled core-shell cocatalysts
Solar energy is perceived as the cleanest energy for 21st century. Sunlight-driven water splitting, which is a promising new technology, provides an approach to directly transform solar energy into hydrogen fuel. Cocatalysts can not only promote electron-hole separation but also provide active sites for H2O adsorption and activation, which makes it widely used in the field of photocatalysis. Among various cocatalysts, platinum has been demonstrated to be a highly active cocatalyst material for water splitting by forming a Schottky junction and providing an active surface, however, high material costs would impede its practical applications. Therefore, the development of high-performance, low-cost photocatalysts is always the ultimate goal for cocatalyst design. Recently, Prof. Yujie Xiong’s group reported the development of atomically controlled Pd@Pt core-shell cocatalysts which can reduce the Pt usage and promote the efficiency of photocatalytic water splitting. This work provides a new way to develop low-costs and high-performance photocatalyts. The research result was published in Angew. Chem. Int. Ed (Angew. Chem. Int. Ed. DOI: 10.1002/anie.201508024) and has been selected as VIP Paper.
The design of atomically controlled Pd@Pt core shell cocatalysts is based on the mechanism of interfacial charge polarization which is developed by their former work. (J. Am. Chem. Soc. 2014, 136, 14650; Angew. Chem. Int. Ed. 2014, 53, 12120)
In this ingenious design, the potential difference between Pd and Pt serves as the driving force for the photogenerated electrons migration across the interface from semiconductor to Pd and Pt. Electron finally accumulated on the exposed Pt surface, which can boost photocatalytic hydrogen evolution reaction. His coworker Prof. Jun Jiang described this process via theoretical simulation. Prof. Junling Lu’s group employed CO as a probe molecule to resolve the electron density of the Pt surface. The results of CO chemisorption examined by diffuse-reflectance infrared fourier transform spectroscopy (DRIFTS) showed that charge specifically accumulated on Pt surface, which confirmed the mechanism.
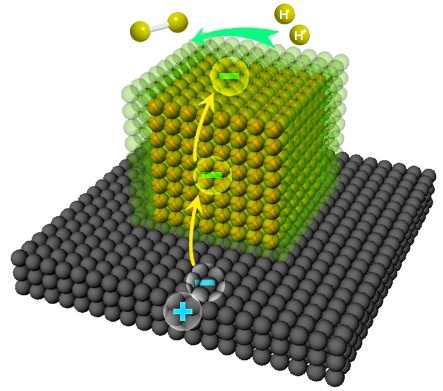
The Mechanism of Photocatalytic Water Splitting via Atomically Controlled Core-Shell Cocatalysts
Interfacial charge polarization puts forward high requirement for the thickness of the Pt shell. Here, the researcher developed a novel approach which can atomically control the thickness of the shell in liquid system. The cost of it is much lower than that of atomic layer deposition. With the development of synthetic technique, the efficiency of photocatalytic water splitting has been remarkably improved by a factor of up to 322 compared to that of bare TiO2 and by a factor of 8.2 compared to that of TiO2-Pt hybrid structures with a comparable Pt {100} surface structure. Meanwhile, the low-priced Pd takes in place of Pt as the core element to reduce the cost. This research proposes a new idea for interfacial engineering which can promote the ability of electron migration in the conversion of solar energy. It also represents a significant step towards the development of high-performance photocatalysts.
This work was financially supported by the 973 Program, the NSFC, the Recruitment Program of Global Experts, the CAS Hundred Talent Program, and the Fundamental Research Funds for the Central Universities.
(School of Chemistry and Materials Science, Collaborative Innovation Center of Chemistry of Energy Materials, Heifei National Laboratory for Physical Science at the Microscale, Hefei Science Center)
http://onlinelibrary.wiley.com/doi/10.1002/anie.201508024/abstract