Cobalt Nanocrystals for Electrocatalytic Hydrogen Generation From Neutral-pH Water
The research group led by Prof. MA Mingming at the University of Science and Technology of China has developed a facile synthetic approach to cobalt-nanocrystal-assembled hollow nanoparticles (Co-HNP), which serve as a highly active and stable electrocatalyst for hydrogen generation from neutral-pH water. This work was published in Angewandte Chemie International Edition on Apr. 29th, 2016 (DOI:10.1002/anie.201601367), and highlighted on the inside cover (DOI: 10.1002/anie.201603780).
Electrolysis of water driven by electricity that is derived from renewable energy sources could provide hydrogen as an ideal energy carrier for clean and sustainable energy technologies. To achieve a high energy conversion efficiency, active catalysts for electrochemical hydrogen evolution reaction (HER) are required. Employing neutral-pH electrolyte solution prevents the undesirable use of strong acids or bases that can lead to environmental and handling issues. Noble metals such as platinum can facilitate HER effectively in a wide pH range, but their scarcity and high cost promote the searching for alternative HER catalysts based on earth-abundant elements.
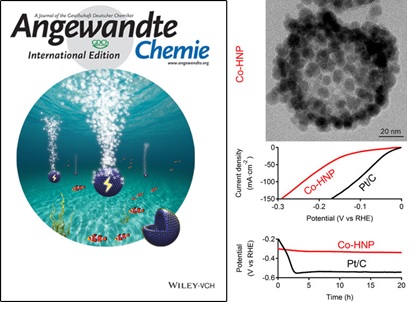
The picture of the inside cover, the structure of Co-HNP and the HER performance (Courtesy of MA Mingming and Angewandte Chemie International Edition)
To address this grand challenge, the researchers led by Prof. MA have developed a "sacrificial template" method to synthesize Co-HNP, which is based on Cobalt, a low-cost earth-abundant metal. The optimized Co-HNP serves as an electrocatalyst for HER from neutral-pH water, which is able to drive large cathodic currents at low overpotentials, and offers stable operation at a heavy-loading condition over an extended period. The performance of Co-HNP for HER from neutral-pH water exceeds that of commercial Pt/C catalyst. These results suggest that non-noble metal nanocrystals assembled in hollow nanostructure are promising for developing highly active and stable electrocatalysts.
This work was financially supported by the NSFC and the Recruitment Program of Global Experts.(CAS Key Laboratory of Soft Matter Chemistry, iChEM, School of Chemistry and Materials Science)
Publication link: http://onlinelibrary.wiley.com/doi/10.1002/anie.201601367/full
http://onlinelibrary.wiley.com/doi/10.1002/anie.201603780/full