Mobility and Reactivity of Oxygen Adspecies on Platinum Surface
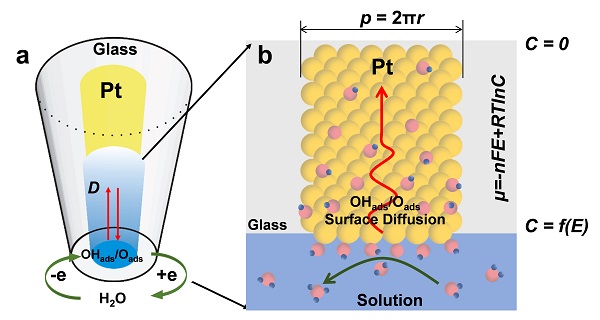
Prof. Zhong-Qun Tian and Dongping Zhan’s group made an important progress on the surface electrochemistry and realized the directly quantitative measurements of surface mobility of adsorbates on platinum catalyst. The adsorption and mobility of oxygen adspecieson platinum (Pt) surface are crucial for the oxidation of surface-absorbed carbon monoxide (CO), which causes the deactivation of Pt catalyst in fuelcells. By employing nanoelectrode and ultramicroelectrode techniques, they have observed the surface mobility of oxygen adspecies produced by the dissociative adsorption of H2O and the surface reaction between the oxygen adspecies and the pre-adsorbed CO on the Pt surface. The desorption charge of oxygen adspecieson a Pt nanoelectrodehas been found to be in proportionto the reciprocal of the square root of scan rate. Using this informaiton, the apparent surface diffusion coefficient of oxgenadspecies has been determined to be(5.61 ± 0.84) × 10-10cm2/s at 25 ºC. The pre-exponential factor D0and the activation energies ΔEd were determined to be 1.62×10-7cm2/s and 13.67 kJ/mol, respectively.During the surface oxidation of CO, two current peaks are observed, which are attributed to direct CO oxidation at the Pt/electrolyte interface and the surface mobility of the oxygen adspecies on the adjcent Pt surface, respectively. These results demonstrate that the surface mobility of oxygen adspecies plays an important role in the anti-poisoning and reactivation of Pt catalyst. Furthermore, the research provides an innovative method for the quantitative measurements of surface mobility for the surface science and heterogonous catalysis. The results were published recently in J. Am. Chem. Soc., DOI: 10.1021/jacs.6b05259.
Paper link:http://pubs.acs.org/doi/abs/10.1021/jacs.6b05259